
This is to show that the subscript applies to the entire polyatomic ion. evidence that cobalt sulfate heptahydrate may indirectly damage DNA by oxidative stress (NTP 1998). If more than one of a particular polyatomic ion is needed to balance the charge, the entire formula for the polyatomic ion must be enclosed in parentheses, and the numerical subscript is placed outside the parentheses. Substance CAS number Molecular formula Cobalt metal Co Cobalt (II) sulfide CoS Cobalt (II) oxide CoO Cobalt (II,III) oxide Co 3O 4 Cobalt. The solubility of calcium phosphate is 114nM in water (x) The solubility of calcium is 341 nM in water (3x) The solubility of. So there are three questions that can be answered here. Information on this page: Condensed phase thermochemistry data References Notes Data at other public NIST sites: X-ray Photoelectron Spectroscopy Database, version 4. Solving for the solubility (x) and noting that K sp (calcium phosphate) 2.07 × 10 33. Use this link for bookmarking this species for future reference. 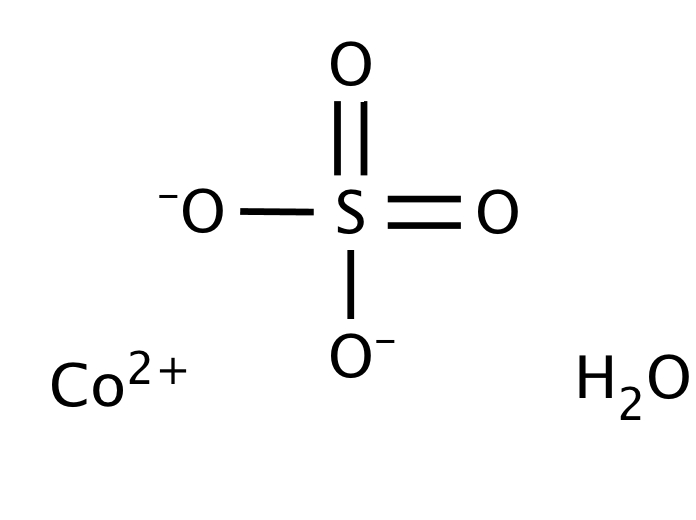 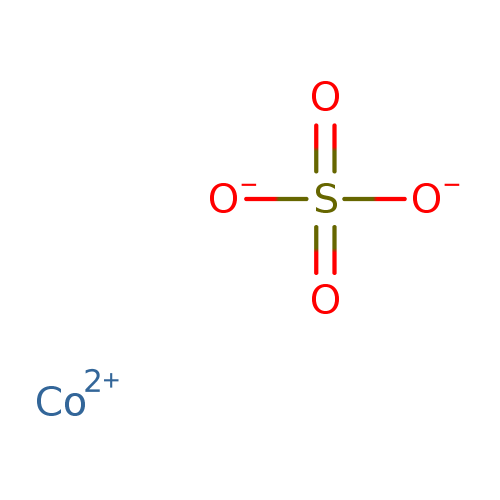
Exposure to this salt can result in the irritation of the eyes, skin, and respiratory tract. Usage of cobalt II sulfate takes place in various electrochemical industries. Moreover, it is a metallic salt that is toxic in nature. It mixes with water as well as sinks in it. The rule for constructing formulas for ionic compounds containing polyatomic ions is the same as for formulas containing monatomic (single-atom) ions: the positive and negative charges must balance. Cobalt(2+) sulfate is a compound of cobalt and sulfate in which the ratio of cobalt (+2 oxidation state) to sulfate is 1:1. Other names: Cobalt sulfate Permanent link for this species. Cobalt II Sulfate refers to an odorless solid that has a rose-pink color.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
